2026 Week 8
What happens at the UPC where you end up with one division deciding on the validity of the patent and another division assessing infringement? Such bifurcated proceedings are the norm in German national patent litigation. But they have been pretty rare at the UPC, where we have seen the Local Divisions routinely decide not to bifurcate where they have a choice.
In some cases, though, bifurcation is forced onto the case by the launch of a standalone revocation action at the Central Division. This week we get to see an example play out, and get a glimpse of how the Court of Appeal may treat the case in due course.
The patent and the dispute
Labrador Diagnostics’ patent is EP 3756767 B1, which is being asserted against a group of bioMérieux companies that manufacture and sell diagnostics equipment. Labrador alleges that bioMérieux’s sales of its VIDAS 3 instrument infringes the patent and bioMérieux denies infringement and contests the patent’s validity.
For those of us following the formalities of UPC practice, the procedural flow of this case is just as interesting as its technical substance. Validity and infringement are bifurcated, with the Milan Central Division (CD) handling validity and the Düsseldorf Local Division (LD) deciding on infringement. Parallel opposition proceedings at the EPO add another layer of complexity. The first-instance hearing at the EPO is scheduled for April 2026.
The main events in the UPC proceedings were:
-
June 2024 – Labrador initiated infringement proceedings against seven bioMérieux entities at the Düsseldorf LD
-
August 2024 – a different bioMérieux entity launches revocation proceedings at the Milan CD
-
September 2024 – the seven bioMérieux entities in the Düsseldorf LD proceedings counterclaim for revocation of the patent as part of their defence to the infringement suit
-
April 2025 – Düsseldorf LD agreed to Labrador’s request to refer the counterclaim for revocation to the Milan CD, to consolidate both strands of the UPC revocation proceedings
-
October 2025 – Milan CD maintains the patent in amended form in their decision bioMérieux v. Labrador
-
January 2026 – Düsseldorf LD finds no infringement of the maintained/amended claims in their decision Labrador v. bioMérieux
-
February 2026 – UPC Court of Appeal (CoA), denies the request from all eight bioMérieux entities to stay the appeal proceedings or extend the deadline for filing statements of grounds of appeal.
Bifurcated proceedings – did either side benefit?
In the revocation proceedings, the Milan CD found that the main request was invalid for added matter and a lack of novelty. The main request consisted solely of two of the originally granted claims, which respectively defined the instrument itself and a method of using it to detect a biological analyte. The third auxiliary request was found to be valid. Claim 1 of the third auxiliary request is reproduced for reference, with the amendments vis-à-vis the corresponding granted claim (claim 9) indicated in bold:
-
An instrument for detecting a biological analyte, comprising:
first means for receiving a device inserted into the instrument, the device comprising:
an array of reagent units, each reagent unit of the array of reagent units for containing a reagent for an assay to detect the biological analyte, and
a sample unit comprising a sample applied by a user; and
second means configured to, with the device received by the first means:
move at least one of a first pipette tip comprising sample or a reagent unit of the array of reagent units relative to the other of the first pipette tip or the reagent unit, for transfer of sample from the sample unit to the reagent unit; and
move at least one of the reagent unit or second pipette tip relative to the other of the reagent unit or the second pipette tip, for transfer of sample from the reagent unit to the second pipette tip, the second pipette tip comprising a capture surface configured to bind with the biological analyte,
a detection assembly for detecting a signal indicative of the presence, absence or con-centration of the biological analyte bound to the capture surface configured to bind with the biological analyte.
The indicated amendments were found to restore validity by overcoming added matter and distinguishing from the prior art. On added matter, the court decided that the granted claim was not directly and unambiguously disclosed in the original (great grandparent) patent application. The granted claim did not require the “sample unit” to be part of the device, whereas the great grandparent disclosed that the sample unit must be present. Moreover, Labrador admitted that it must be present, as now specified in the amended claim.
On novelty, the Milan CD determined that a prior art device disclosed in ‘Clark’ satisfied the instrument as defined by the granted claim. The Clark device used a first pipette tip to move reagent and sample into a “MEIA cartridge”, which is pictured below in Figure 55 from Clark:
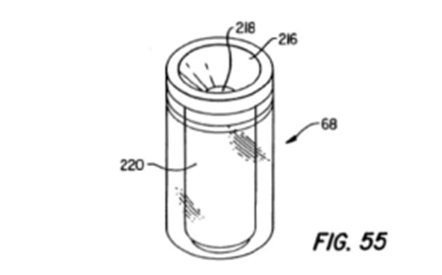
The Milan CD decided that the MEIA cartridge shown in Figure 55 could be considered to be a “second tip” as specified in the granted claim, but was not considered to be a second pipette tip as specified in the claims of the third auxiliary request. Therefore, specifying the second tip as a pipette tip conferred novelty over Clark – and the court also found that the skilled person would not arrive at the claimed configuration in an obvious way in view of the prior art.
It is notable that another piece of prior art advanced by bioMérieux was a publication about their legacy VIDAS systems from before the patent’s priority date. The Milan CD did not consider this publication to be novelty destroying because the legacy systems required the user to manually pipette the sample into the reagent strips. The court held that:
The skilled person will understand that “a second means configured to (…) move” a pipette tip or a reagent unit refers to a system which implements an automated action and excludes any manual operation. Only technical means may be patented, manual operations being out of the scope of patentability.
The Düsseldorf LD’s finding of non-infringement did not focus on the second tip being a pipette. Rather, the court decided that the VIDAS 3 instrument did not have a first means for receiving a device and a distinct device that is inserted.
The Düsseldorf LD took the relevant claim construction from the validity proceedings, noting that:
the Claimant failed to show that the challenged embodiment is an instrument with first means for receiving a device inserted into the instrument which comprises an array of reagent units and a sample unit comprising a sample applied by the user.
At the oral hearing, Labrador advanced a new line of argument, which took the sub-racks on the rack structure as part of the device to hold the sample vial/tube as being the sample unit for receiving the sample applied by the user, to try to show that the VIDAS 3 embodiment had a device element that is distinct from the sample unit. While not committing to admit this late-made argument into the proceedings, the court said it was doubtful that this interpretation would bring the alleged infringement into the scope of the claim because the sample vial/tube did not physically receive the sample unit.
The key point about the bifurcation was that the issue of validity was determined before infringement. This is unlike normal German national patent litigation, in which infringement will usually be determined some time before validity. On this basis, and in view of the attention paid by the Düsseldorf LD to the claim interpretation used by the Milan CD, there was no advantage given to the patentee by bifurcation.
EPO opposition proceedings
It is interesting to note that in the preliminary opinion of the EPO opposition division, a particular “dual-strip” embodiment of the legacy VIDAS instrument is considered to be novelty destroying, including for the auxiliary request upheld by the Milan CD.
In response, the patentee has recently filed a large number of further auxiliary requests with new amendments seeking to address the points raised in the preliminary opinion. If the new amendments are admitted at the upcoming EPO oral proceedings, this means that the EPO will be deciding on different claims to the UPC. If the patent is then maintained in a different amended form to the UPC, we are in uncharted territory – which claims will be in force in the UPC territory? Slightly amazingly, the answer is not yet clear.
The preliminary opinion of the EPO opposition division was presented to the Düsseldorf LD, but their view was that the question of validity was not theirs to assess in a bifurcated case.
What about equivalence?
Labrador raised the question of infringement by equivalence. Although raised late (and therefore possibly not admissible), the Düsseldorf LD dismissed this on the basis that the alleged equivalent does not perform essentially the same function as the device of claim 1.
So in this case, DoE did not bite. But, presumably, if it did need to be considered properly, at least part of the test for equivalence to be applied by the UPC would need to check for obviousness of the alleged infringement over the prior art. This would need a flavour of validity assessment, even in a bifurcated case.
UPC Court of Appeal refuses to stay or delay the appeal
Most recently, we have a 17 February 2026 order from the UPC CoA, dealing with appellate aspects of the case.
In requesting a stay, bioMérieux argued that the upcoming EPO opposition hearing means that a rapid decision on validity is expected, which gives the CoA discretion to stay the UPC proceedings. They argued that the preliminary opinion of the EPO Opposition Division indicates that it is “very likely” that the OD will revoke the patent. They also requested a three month extension of the deadline for filing their Statement of grounds of appeal against the Milan CD’s decision to maintain the patent, to align the deadline with the deadline for Labrador to appeal the finding of non-infringement from the Düsseldorf LD.
The CoA refused to stay the proceedings, noting that the OD proceedings will be open to appeal and that, even without a stay, the parties will have ample opportunity to comment on the EPO opposition decision in the UPC appeal proceedings:
Leaving aside the fact that the decision by the OD will be open to appeal, the OD’s decision is non-binding and can be taken into account by bringing it to the attention of the Court of Appeal at a later stage of the proceedings. A stay is not necessary to achieve this.
Regarding the request to delay the deadline for filing the Statement of grounds of appeal, the CoA refused, noting that filing this Statement on the same day as a possible future Statement of grounds of appeal against the Düsseldorf LD’s finding of non-infringement is “not necessary to establish such a link between validity and infringement”.
The CoA do say something else interesting, though. Noting that it would be efficient to align the revocation and infringement proceedings, they comment that it would be possible to hear the appeals in the revocation and infringement proceedings together. If they do that, we will be toasting a new term in UPC practice: de-bifurcation on appeal.
Eliot is a highly valued partner at Mewburn Ellis and a key driver within the Life Sciences team. With a reputation for driving complex projects from inception to completion, he works with a diverse range of clients, from innovative startups to multinational corporations worldwide. Eliot handles a diverse client portfolio spanning the life sciences and MedTech sectors. A skilled patent prosecutor, Eliot also has wide experience of drafting patent applications on breakthrough technologies, as well as leading offensive and defensive opposition proceedings post-grant. Eliot is also experienced in handling Freedom to Operate projects and in performing due diligence, which have led to the successful completion of high value transactions and investment rounds.
Email: eliot.ward@mewburn.com


.png)